Laboratory Research
An Overview
Research in the Hirbe lab is focused on better understanding the biology of sarcomas, rare tumors of bone and soft tissue. There are over 100 different types of sarcomas all with a different underlying biology. We aim to utilize genomic information from patient sarcoma samples coupled with mouse models to better understand the development and progression of these cancers. Based on identification of mutated DNA repair pathways in a number of sarcomas (MPNSTs, rhabdomyosarcomas, pleiomorphic sarcomas, and osteosarcomas), there is an ongoing project aimed at evaluation of therapies targeting this defect for treatment of bone and soft tissue sarcomas.
We also have a special interest in better understanding the development and spread of a particular soft tissue sarcoma, the malignant peripheral nerve sheath tumor (MPNST), an aggressive soft tissue sarcoma that occurs at an increased frequency in patients with the Neurofibromatosis Type 1 (NF1) tumor predisposition syndrome. Approximately 13% of individuals with NF1 will develop MPNSTs during young adulthood. Currently, there are no predictive biological markers of disease progression, few therapeutic options, and dismal survival. Leveraging whole exome sequencing methods, we have identified several genes mutated in MPNSTs that may play a role in progression of these tumors.
Overview of NF1 Tumor Suppressor Gene Presentation
In understanding sarcoma biology, the Hirbe lab utilizes 3 main tools:
Cell Culture Models:
In the Hirbe Lab we use cultured cell lines to perform genetic manipulations and biochemical work in order to understand the molecular pathways underpinning sarcoma biology. These cell lines are then used in in vivo models to look at the roles these alterations play in cancer progression.
Patient Samples and Clinical Features:
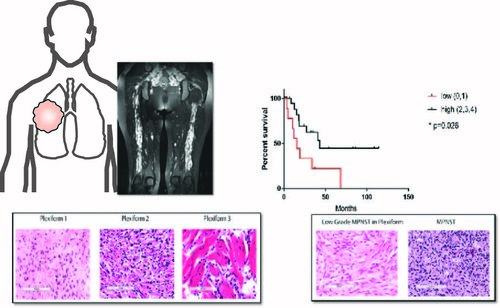
Hirbe Lab uses clinical data and patient samples to identify diagnostic and prognostic
markers to help provide patients with better understanding of their disease and make better predictions of progression.
Patient Derived Xenografts (PDX):
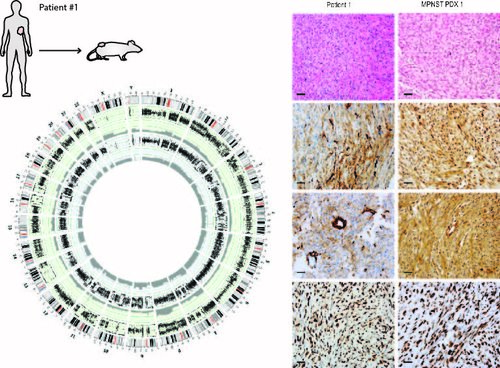
In the Hirbe Lab we are building a suite of tools with
comparative genomic information, expression profiles, and histology for drug discovery that represents the clinical diversity within these of sarcomas.
We are working in collaboration with the Pratilas lab at Johns Hopkins University to build a more comprehensive set. The goal of Dr. Pratilas’ project is to establish a comprehensively annotated biorepository of tissue and blood fractions for use in NF1 research. For more information about their work to bank NF1-associated tumors, including plexiform neurofibromas, MPNST, and blood fractions from patients with NF1, please click the button below.